Services Regulatory Submissions Management
Regulatory Submissions Management
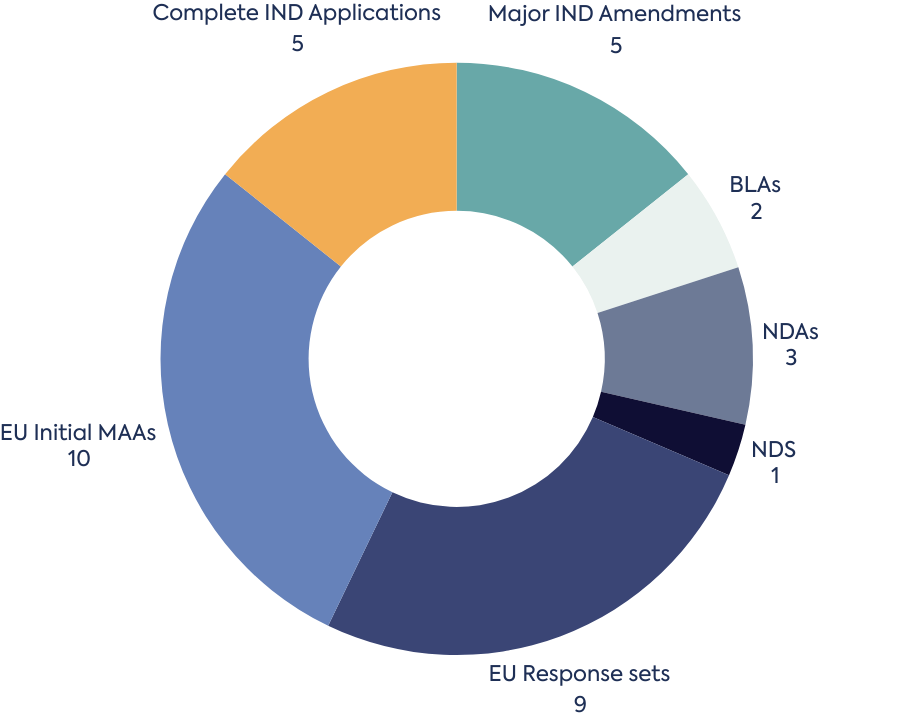
At DLRC, our Regulatory Operations team bring over 75 years of combined experience in managing regulatory submissions across global regions. We offer a comprehensive, end-to-end service tailored to your needs. From strategic submission planning to electronic Common Technical Document (eCTD) compilation, our team ensures that every submission meets the highest quality and compliance standards.